.png)
Indeed, these paclitaxel-induced increases in peripheral cytokines are significantly correlated with increased fatigue in cancer patients. Taxane drugs, including paclitaxel, are associated with increased circulating concentrations of the pro-inflammatory cytokines interleukin (IL)-1β, IL-6, and tumor necrosis factor (TNF)-α in cancer patients and preclinical models, , ]. Systemic inflammation resulting from immune cell signaling triggered by chemotherapy-induced cell death, is posited as a primary contributor to weight loss, anorexia, and fatigue. Compared to other chemotherapeutic drugs ( e.g., cyclophosphamide, doxorubicin, 5-fluorouracil, methotrexate ), the causal role and underlying mechanisms of taxane drugs in physiological and behavioral toxicities is poorly understood. One common chemotherapeutic drug, paclitaxel, is a taxane agent that binds to microtubules to prevent proper mitotic spindle formation and is a “mainstay” treatment for breast, ovarian, and other cancers (reviewed in ). These “sickness behavior” side effects of chemotherapy persist during and following treatment resulting in frequent patient reports of severely decreased quality of life.
Cancer-related fatigue is different from other forms of fatigue as it is neither caused by increased activity levels nor restored by rest, and is comprised of central (motivational) and peripheral (metabolic and muscle-driven) components. Nevertheless, these drugs also kill dividing healthy cells and thereby contribute to a wide range of well-documented chemotherapy-related toxicities, including severe weight loss, anorexia, and fatigue. The present findings demonstrate the benefits of exercise on faster recovery from paclitaxel-induced body mass loss and deficits in melanocortin signaling and suggests the development of therapies targeting the melanocortin pathway to reduce paclitaxel-induced weight loss.Ĭhemotherapy drugs are instrumental components of cancer treatment that kill neoplastic tumor cells.
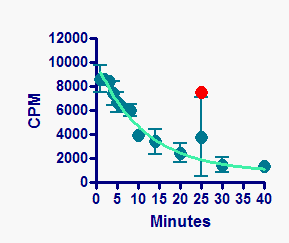
Body mass recovery was associated with the wheel running-induced recovery of body composition, paclitaxel-induced alterations to hypothalamic melanocortin signaling, and associated peripheral circulating hormones (ghrelin and leptin). Exercise intervention did not ameliorate fatigue or inflammation, but hastened recovery from paclitaxel-induced weight loss.

Circulating interleukin (IL)-6 and hypothalamic Il1b gene expression remained elevated in chemotherapy-treated mice at least 3 days after the last dose. Paclitaxel treatment also transiently decreased locomotion (open field test), voluntary wheel running, home-cage locomotion, and core body temperature without affecting motor coordination (rotarod task). Body mass and food intake decreased following six doses of chemotherapy treatment relative to vehicle controls, lasting less than 5 days after the last dose. Furthermore, we sought to identify the extent to which voluntary exercise (wheel running) attenuates paclitaxel-induced sickness behaviors and underlying central pathways. Here, we tested the longitudinal relationship between a clinically-relevant paclitaxel regimen, inflammation, and sickness behaviors (loss of body mass, anorexia, fever, and fatigue) in adult, female mice. However, the mechanisms underlying taxane chemotherapy-induced side effects, and effective treatments to ameliorate them, are not well-established. While chemotherapy remains a common cancer treatment, it is associated with debilitating side effects ( e.g., anorexia, weight loss, and fatigue) that adversely affect patient quality of life and increase mortality.

